Amines NCERT PDF from Class 12 Chemistry is an essential chapter that introduces students to nitrogen-containing organic compounds. Amines play a crucial role in daily life and industrial chemistry, as they form the basis for several pharmaceuticals, dyes, and polymers. This chapter builds a strong understanding of organic functional groups, naming conventions, physical and chemical properties, and preparation methods of amines.
In this comprehensive guide, you will explore different types of amines—primary, secondary, and tertiary—along with their methods of preparation and reactions such as alkylation, acylation, and diazotization. Understanding amines is not only vital for board exams but also for Competitive Exams like JEE and NEET, where reaction mechanisms and applications often appear.
By the end of this lesson, you will know How To classify amines, write their IUPAC names, understand basicity trends, and apply key equations like \(pK_b = -\log K_b\) for numerical and conceptual questions. Let’s dive deeper into the key areas of the NCERT Amines Class 12 PDF and break down the chapter conceptually and visually.
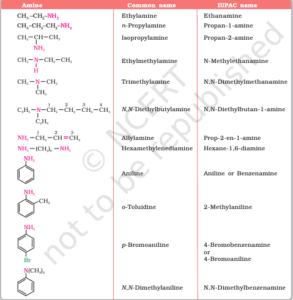
Table of Contents
- Classification and Structure of Amines
- Preparation Methods of Amines
- Chemical Properties and Reactions
- Uses and Industrial Importance
- FAQs
Classification and Structure of Amines
Types of Amines Based on Alkyl Substitution
| Type of Amine | General Formula | Example | Nature |
|---|---|---|---|
| Primary (1°) | R–NH2 | Methylamine (CH3NH2) | One alkyl group attached |
| Secondary (2°) | R2NH | Dimethylamine ((CH3)2NH) | Two alkyl groups attached |
| Tertiary (3°) | R3N | Trimethylamine ((CH3)3N) | Three alkyl groups attached |
| Quaternary Ammonium Salts | R4N+X– | Tetramethylammonium chloride | Ionic, used in surfactants |
The classification of amines depends on the number of carbon-containing groups attached to the nitrogen atom. In primary amines, only one alkyl or aryl group is attached; in secondary and tertiary, two and three respectively. The electron density on nitrogen determines their basicity. Tertiary amines are less soluble in water compared to primary amines due to reduced hydrogen bonding.
- The nitrogen atom in amines is sp3 hybridised and has a lone pair of electrons, which gives amines their characteristic basic nature. The molecular geometry around nitrogen is pyramidal, leading to bond angles of approximately \(108^{circ}\). This geometry and lone pair repulsion significantly affect reactivity and intermolecular interactions.
Preparation Methods of Amines
Common Laboratory and Industrial Synthesis Routes
| Method | Reactants | Products | Equation |
|---|---|---|---|
| Ammonolysis of Alkyl Halides | Alkyl halide + NH3 | 1°, 2°, 3° Amines | \(R–X + NH_3 ightarrow R–NH_2 + HX\) |
| Reduction of Nitro Compounds | Nitrobenzene + H2 | Aniline | \(C_6H_5NO_2 + 3H_2 ightarrow C_6H_5NH_2 + 2H_2O\) |
| Gabriel Phthalimide Reaction | Phthalimide + Alkyl halide | Primary amine | \(C_6H_4(CO)_2NH + R–X ightarrow RNH_2\) |
| Hofmann Bromamide Reaction | Amide + Br2 + KOH | Primary amine (one carbon less) | \(RCONH_2 + Br_2 + 4KOH ightarrow RNH_2 + K_2CO_3 + 2KBr + 2H_2O\) |
The preparation of amines is a cornerstone of organic synthesis. Ammonolysis provides a sequence of amines depending on reaction control, while the Gabriel phthalimide synthesis is best suited for primary amines because it avoids multiple substitutions. Reduction of nitro compounds, such as converting nitrobenzene to aniline, remains one of the most common industrial processes.
Hofmann’s bromamide reaction is especially important for reducing carbon count, which helps students understand how reaction mechanisms can alter chain length. Each synthesis type highlights how electron-donating or withdrawing groups affect the final yield. It’s also important to compare yields under different conditions to understand kinetic vs thermodynamic control in organic synthesis.
Chemical Properties and Reactions
Key Reactions Demonstrating Basicity and Reactivity
| Reaction Type | Example Reaction | Observation |
|---|---|---|
| Acylation | \(RNH_2 + CH_3COCl ightarrow RNHCOCH_3 + HCl\) | Forms amides |
| Alkylation | \(RNH_2 + R’X ightarrow R_2NH + HX\) | Produces higher amines |
| Diazotization | \(C_6H_5NH_2 + HNO_2 + HCl ightarrow C_6H_5N_2^+Cl^- + 2H_2O\) | Formation of diazonium salt |
| Carbylamine Reaction | \(RNH_2 + CHCl_3 + KOH ightarrow RNC + 3KCl + 3H_2O\) | Unpleasant odor confirms primary amines |
The chemical behavior of amines largely stems from the lone pair on nitrogen, which makes them nucleophilic and basic. In acylation and alkylation, the nitrogen attacks electrophiles, forming stable derivatives. The carbylamine reaction serves as a distinguishing test for primary amines due to the characteristic foul smell of isocyanides produced.
One of the most crucial reactions is diazotization, where an aromatic amine like aniline reacts with nitrous acid to form a diazonium salt. This intermediate enables multiple coupling reactions to create azo dyes—a real-world application linking chemistry to textile industries. Students should remember that diazotization occurs at temperatures below \(5^{circ}C\) since diazonium salts are unstable above that.
Uses and Industrial Importance
Applications of Amines in Everyday Life and Industry
| Application Area | Examples | Significance |
|---|---|---|
| Pharmaceuticals | Ephedrine, Chlorpromazine | Act as active ingredients in drugs |
| Dyes | Aniline-based azo dyes | Used in textile and printing industries |
| Polymers | Nylon and Polyurethane | Derived from diamines |
| Agriculture | Herbicides like Atrazine | Improves crop productivity |
Amines are building blocks for countless organic and biochemical compounds. Aniline derivatives are the backbone of the dye industry, giving us bright, long-lasting colors. Diamines are used in polymer synthesis like nylon, illustrating the interdisciplinary connection between organic chemistry and materials science.
In the medical field, amine derivatives are part of vital drug structures such as adrenaline and antihistamines. These applications show the relevance of NCERT concepts beyond exams—connecting molecular structures to real-world innovations. For instance, tertiary amines act as corrosion inhibitors and catalysts in industrial reactions, showcasing their versatility and importance.